
Demonstrated Efficacy in Advanced ACC: Lysodren® Plus Combination Chemotherapy (EDP or etoposide, doxorubicin and cisplatin) in Metastatic ACC2*
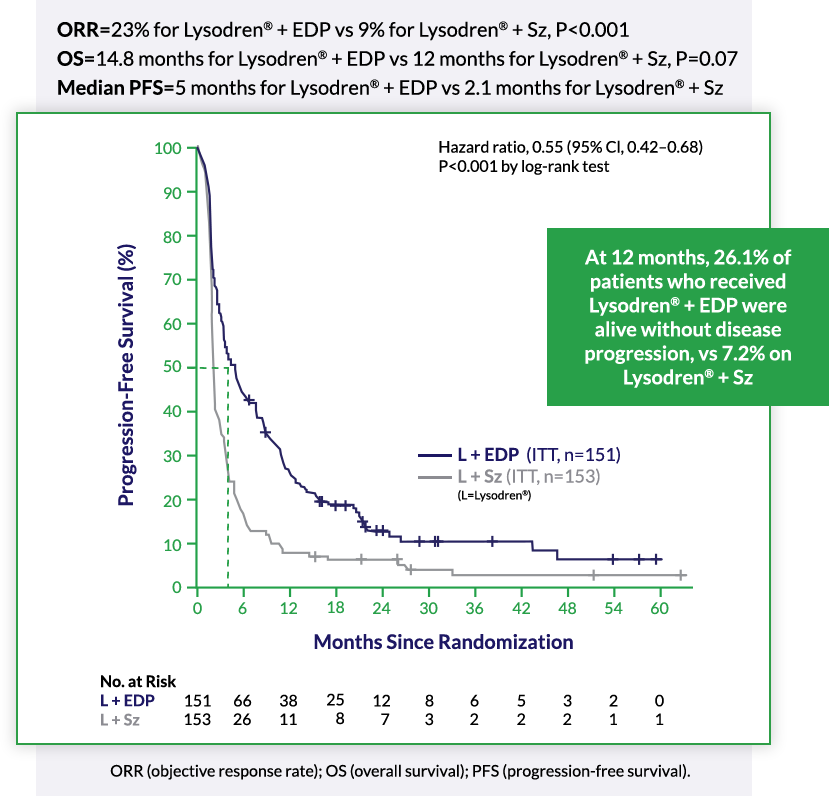
*The above FIRM-ACT (First International Randomised Trial in Locally Advanced and Metastatic Adrenocortical Carcinoma Treatment) Trial is an international phase 3 trial in metastatic ACC which studied the combination of Lysodren® + EDP (etoposide, doxorubicin and cisplatin) (ITT, n=151) vs treatment with Lysodren® + Sz (streptozotocin) (ITT, n=153). PFS and ORR were significantly improved with Lysodren® + EDP vs Lysodren® + Sz. Primary end point of the study: overall survival. Secondary end points: progression-free survival, tumor response, and quality of life.
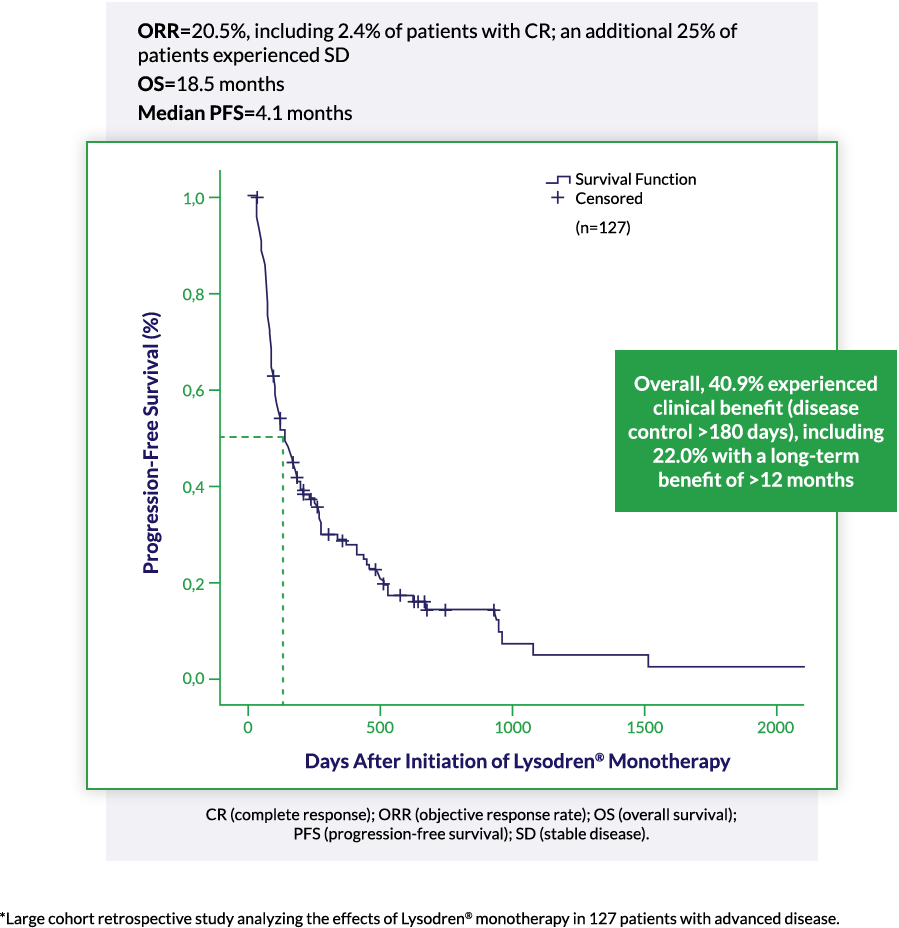
Demonstrated Efficacy in Advanced ACC: Lysodren® Monotherapy in Advanced ACC3*
Endocrinology and Oncology Guidelines Recommend Lysodren® for the Treatment of Advanced ACC.
NCCN Clinical Practice Guidelines in Oncology V1.0 20234
LOCOREGIONAL UNRESECTABLE/METASTATIC ACC
Preferred Regimens
• Carboplatin + etoposide ± doxorubicin ± mitotane (Lysodren®)
• Cisplatin + etoposide ± doxorubicin ± mitotane (Lysodren®)
Other Recommended Regimens
• Mitotane (Lysodren®) monotherapy
• Pembrolizumab ± mitotane (Lysodren®)
ESE-ENSAT Clinical Practice Guidelines 20185*
ADVANCED ACC NOT AMENABLE TO RADICAL RESECTION
mitotane (Lysodren®) is the treatment of choice as either mitotane (Lysodren®) monotherapy or mitotane (Lysodren®) + EDP depending on prognostic parameters (+++O)
*The quality of evidence behind the recommendations is classified as very low (+OOO), low (++OO), moderate (+++O), and strong (++++).
ESMO-EURACAN Clinical Practice Guidelines 20206
ADVANCED/METASTATIC ACC
mitotane (Lysodren®) monotherapy or in combination with chemotherapy
The 2 main biological effects Lysodren® exhibits are:
1
Direct cytotoxic effect on adrenocortical cells1
2
Decrease in the production of steroids, including cortisol, androgens, and dehydroepiandrosterone from the adrenal cells1
In particular, its metabolites inhibit several enzymes in the adrenocortical steroidogenesis pathway, mainly at the level of the cholesterol side-chain cleavage enzymes CYP11A1 and CYP11B1.7 By inhibiting CYP11A1 and CYP11B1 and together with its adrenolytic effects, Lysodren® may lead to some control of hormone secretion,8 which may further benefit patients with functioning ACC.

The precise mechanism of its antitumor action is not fully understood.

Lysodren provides links to web sites of other organizations in order to provide visitors with certain information. A link does not constitute an endorsement of content, viewpoint, policies, products or services of that web site. Once you link to another web site not maintained by Lysodren, you are subject to the terms and conditions of that web site, including but not limited to its privacy policy.
Click the link above to continue or CANCEL