Lysodren® is available as 500 mg tablets.
The recommended initial dose of Lysodren® is 2000 mg to 6000 mg orally, divided in 3 or 4 doses per day. Dosage may vary based on the medical needs of each patient.

Lysodren® should be taken with food; administration with high-fat food enhances absorption. Timing of the dose relative to meals must be consistent.
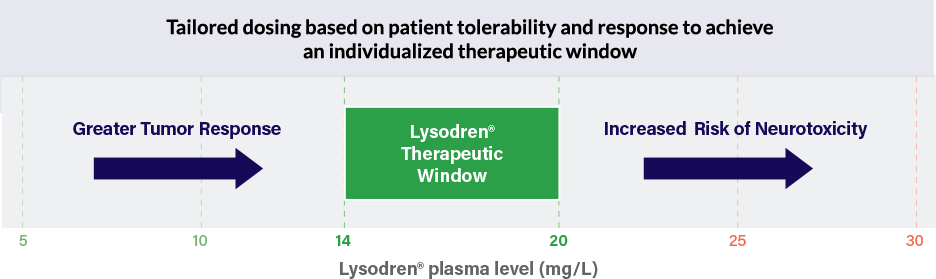
Lysodren® has a well-documented safety profile. The most common adverse events include GI, nervous system, metabolism, or endocrine disorders.2
Specifically:
For a full list of possible side effects, please see the Lysodren® Prescribing Information.
Because of the adrenolytic effects of Lysodren®, glucocorticoid replacement (eg, hydrocortisone/cortisone acetate) is recommended in patients treated with Lysodren® (except those with functioning tumors with ongoing cortisol excess).3
Due to increased steroid clearance and increase in cortisol-binding globulin, at least twice the standard replacement dose is usually required.3


Lysodren provides links to web sites of other organizations in order to provide visitors with certain information. A link does not constitute an endorsement of content, viewpoint, policies, products or services of that web site. Once you link to another web site not maintained by Lysodren, you are subject to the terms and conditions of that web site, including but not limited to its privacy policy.
Click the link above to continue or CANCEL